
"Density Formula and Concentration Inequalities with Malliavin Calculus." Electron. We also explain how to derive concentration inequalities for $Z$ in our framework. Among several examples, we provide an application to the (centered) maximum of a general Gaussian process. Here, thanks to $G$, density lower bounds can be obtained in some instances. The use of $G$ was first discovered by Nourdin and Peccati (PTRF 145 75-118 2009 MR-2520122), in the context of rates of convergence in law. The formula is based on an auxilliary random variable $G:= _H$, where $L$ is the generator of the so-called Ornstein-Uhlenbeck semigroup. The main advantage of this formula is that it does not refer to the divergence operator $\delta$ (dual of the Malliavin derivative $D$). It is worth remembering what density is in the scientific world.We show how to use the Malliavin calculus to obtain a new exact formula for the density $\rho$ of the law of any random variable $Z$ which is measurable and differentiable with respect to a given isonormal Gaussian process. Remember that what we say in common is not always reflected in the scientific world. This follows on from the formula for density. Of course, the correct answer is that their density is lower than that of water (they are lighter and, therefore, float freely on water). Now you must know why ice and wood can float on water. 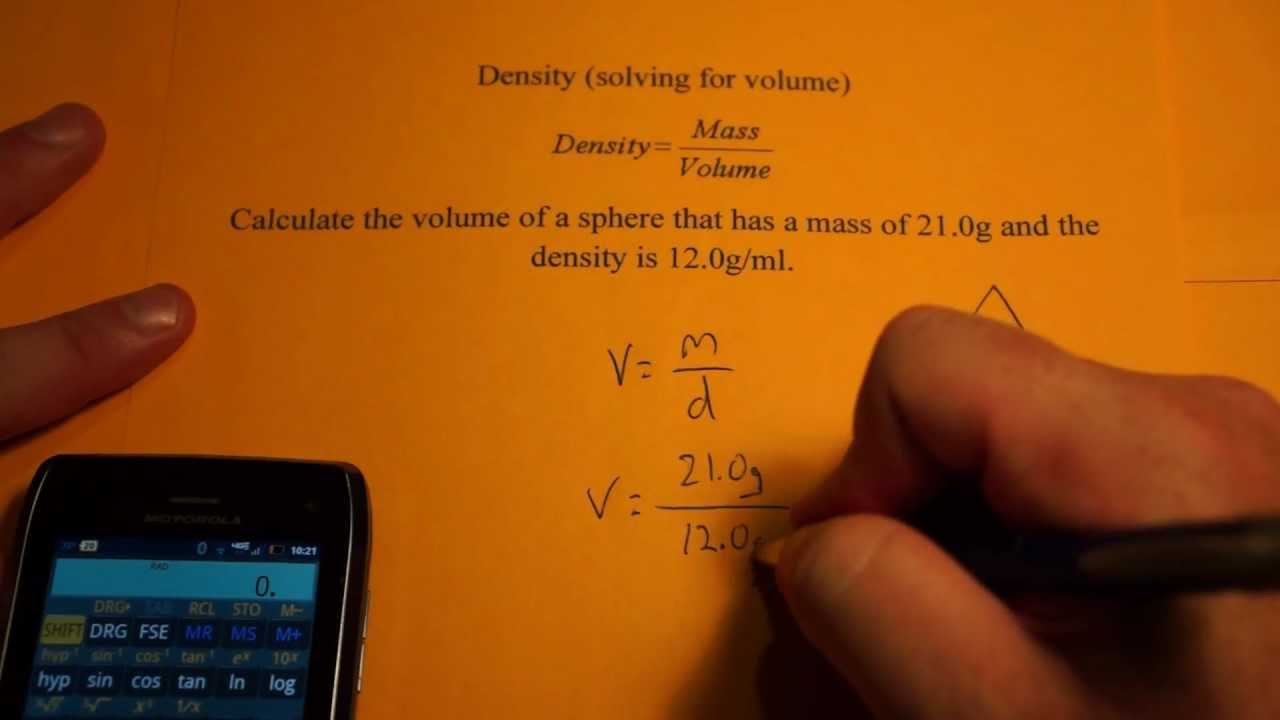
The easiest way to measure density is in the laboratory – simply weigh the material of interest and then immerse it in water and calculate the increase in volume in the container. In principle, it doesn't matter much for solids or even liquids, but you will easily notice the changes when it comes to gases. Density can change with the same mass and volume – it depends on temperature and pressure. Finding the density of an object requires the use of a density formula. If we know the density, we can calculate the mass of a body provided that we know its volume or its volume when we know its mass. When trying to understand what density is, it is useful to remember a few basic things – firstly, in physics, density is not a fluidity or viscosity, but defines the heaviness of a material. 
The formula for density is one of the simplest formulas. 
There are two basic units of density – kilogram per cubic metre and gram per cubic centimetre. The density formula is quite universal in the scientific world. This principle works not only in physics, but also in chemistry. The formula for density can be calculated by knowing the mass and volume of a material – simply divide one by the other. How do you calculate density? What is the formula for density? And one of the densest materials (and the most valuable) is gold. Wood, for example, can also be lighter than water. Because ice floats on water, hence it has a lower density. The best way to understand the concept of density is to use the example of water and ice. Units of Density The units of density depend on the units of mass and volume used in the formula. In other words, it is the ratio or proportion of mass to volume. The formula for calculating the density of a substance is: Density Mass/Volume d. The definition of density is not difficult to understand: it is simply the mass of a substance per unit volume. Today, the subject of this article will be density. Physics surrounds us whether we are aware of it or not.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
